Save and Support Life.
Pioneering cardiopulmonary support systems that push the boundaries of medical technology. With over a decade of innovation, we develop and manufacture life-critical devices trusted by clinicians worldwide.
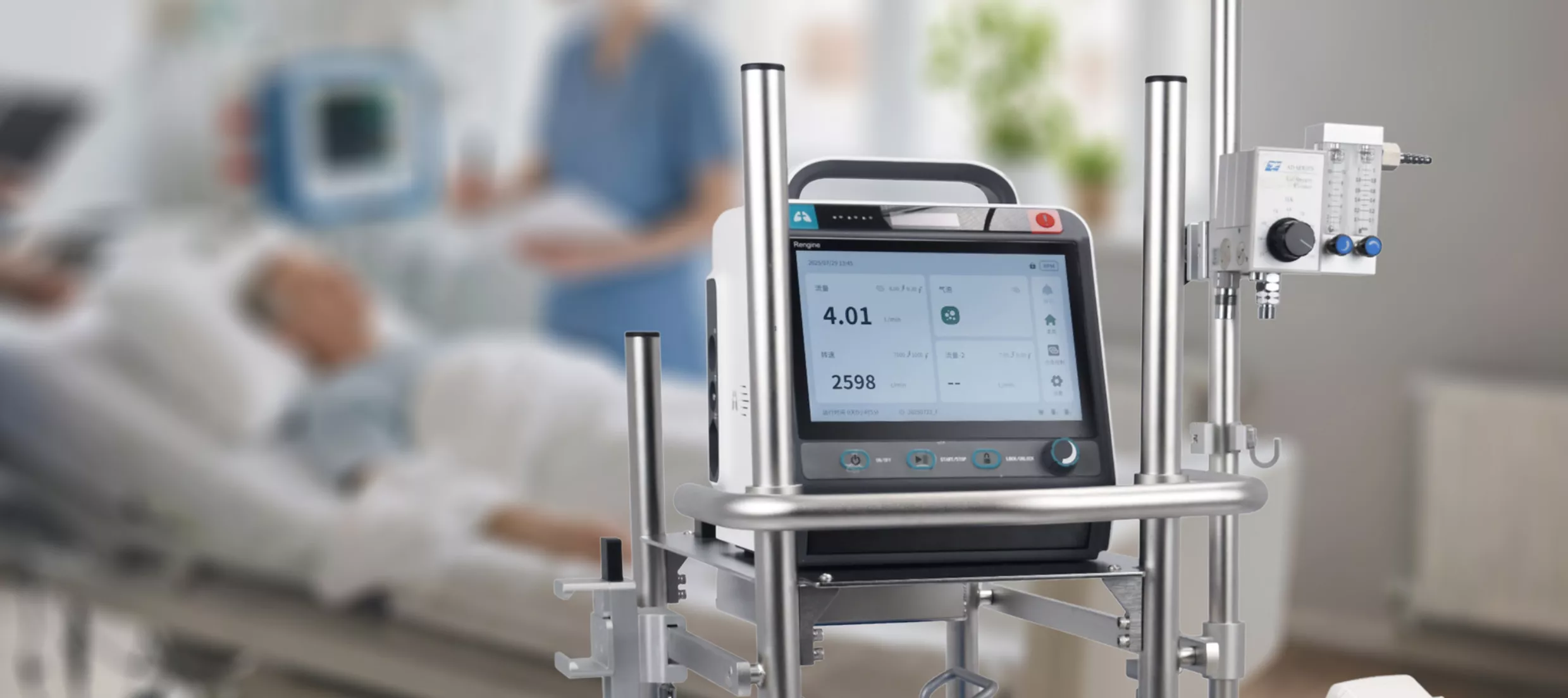
Pioneering cardiopulmonary support systems that push the boundaries of medical technology. With over a decade of innovation, we develop and manufacture life-critical devices trusted by clinicians worldwide.


LifeShield Medical is a globally active developer and manufacturer of advanced medical devices specializing in cardiopulmonary support systems. Our expertise is rooted in academic research and refined through over a decade of clinical and industrial application.
With operations in Germany and China, we combine world-class engineering with agile manufacturing to deliver life-critical solutions that meet the highest international standards.
Experienced leaders driving innovation in cardiopulmonary medical technology.



Our portfolio of cardiopulmonary devices is designed to support clinicians in delivering the best possible patient outcomes.
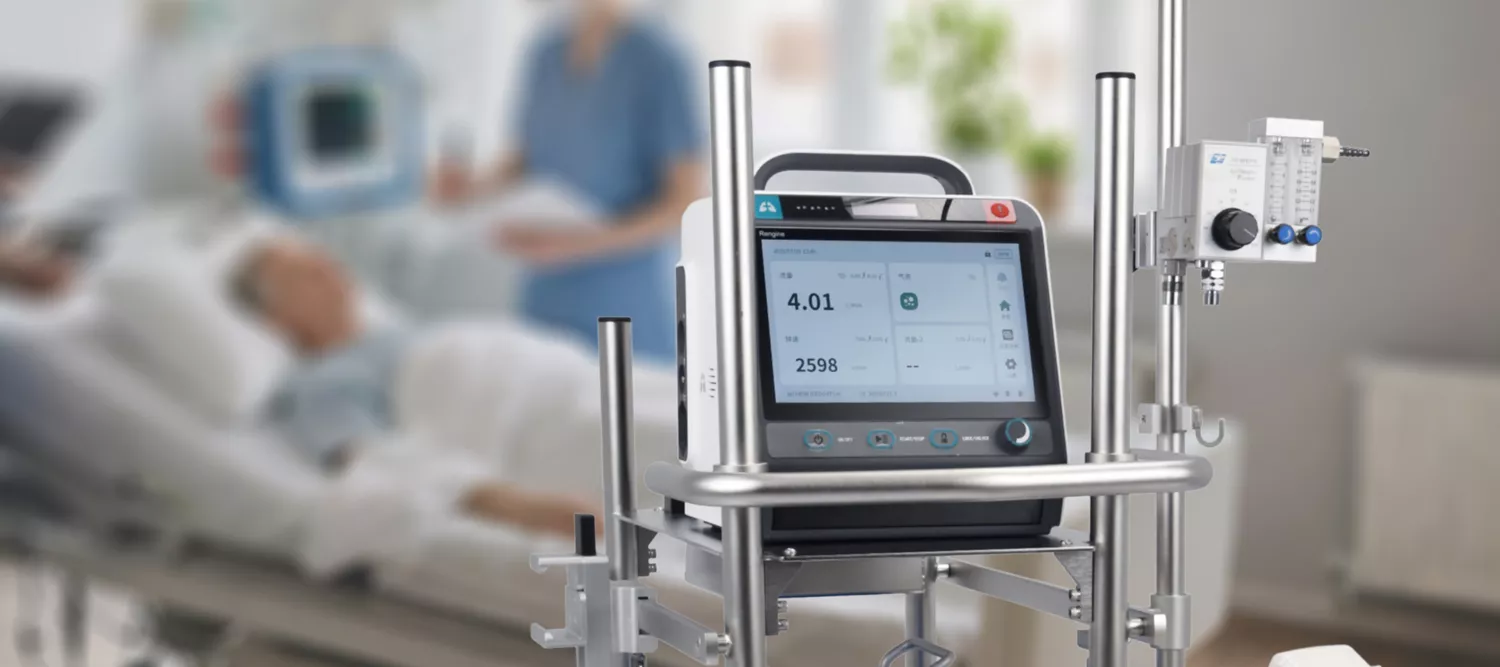
State-of-the-art circulatory support systems optimized through simulation science and validated in clinical settings worldwide.
Learn More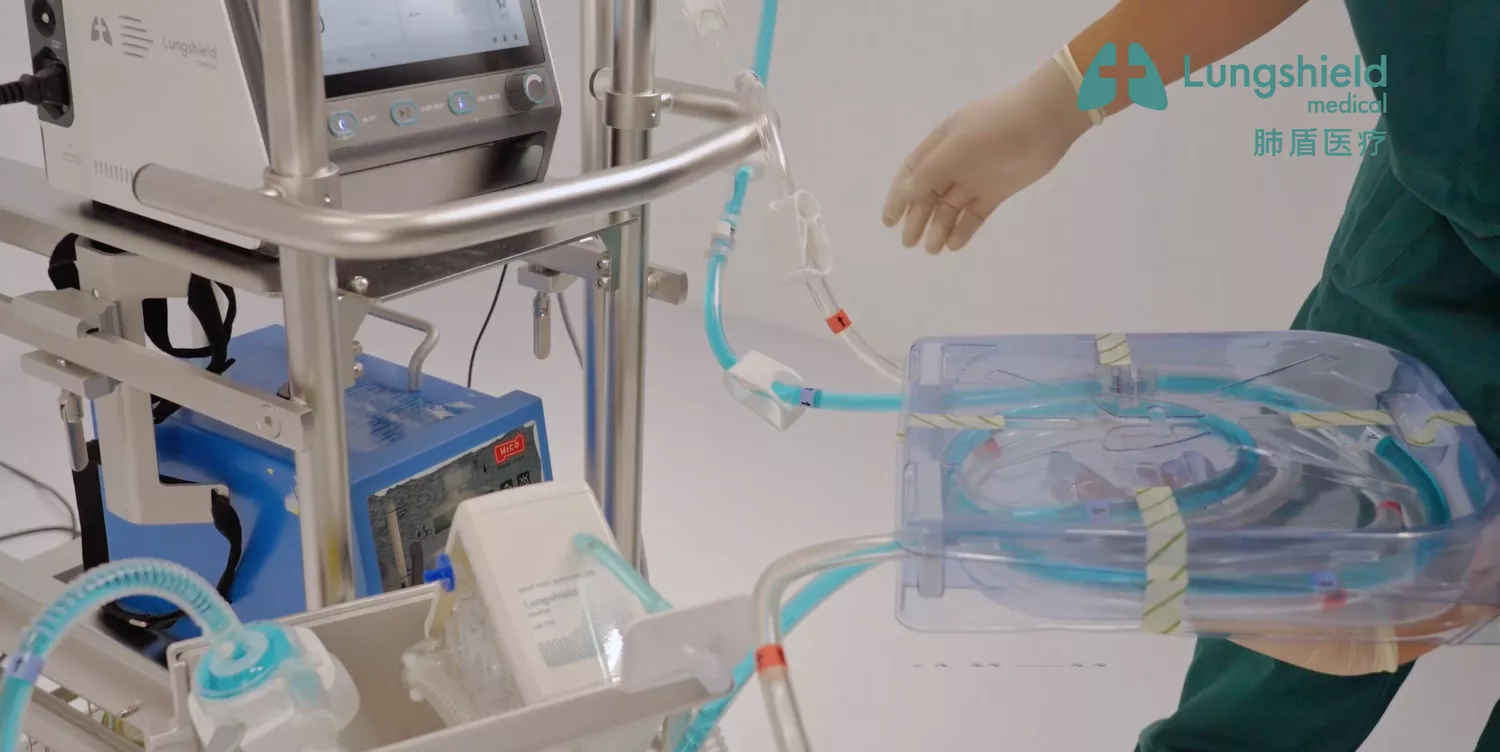
Extracorporeal membrane oxygenation solutions engineered for reliability and performance in critical care environments.
Learn More
Precision-engineered heart-lung machines designed for the highest levels of safety and clinical ease of use.
Learn MoreFrom concept through to certified product, we deliver end-to-end medical device development grounded in simulation science.
Full-cycle medical device development from initial concept to market-ready product.
Advanced computational modeling to optimize device performance and patient outcomes.
ISO 13485 certified manufacturing with rigorous quality management systems.
Translating clinical needs into device specifications validated through real-world testing.
Quality is at the core of everything we do. Our integrated quality management system is certified to ISO 13485 and verified by TÜV Süd, ensuring that every product we develop and manufacture meets the most stringent international regulatory requirements.
From design controls through post-market surveillance, our processes are built to deliver safe, effective, and reliable medical devices.
Learn More About Quality
Strategically positioned in Europe and Asia to serve clinical partners worldwide.
European headquarters and primary R&D center. Home to 40+ engineers, scientists, and regulatory specialists driving innovation in cardiopulmonary care.
Tempelhofer Str. 16, 52068 Aachen, Germany
+49 (0)241 412-510-71
30+ team members at our partner facility Lungshield Medical, providing scalable manufacturing capacity and regional market access across Asia.
Stay up to date with the latest developments from LifeShield Medical.

Growing demand for cardiopulmonary support systems drives expansion of our Aachen facility.

Meet our team and explore our latest innovations at the world's leading medical trade fair.

How computational modeling is reshaping the development of next-generation ECMO systems.
Whether you're exploring cardiopulmonary solutions or seeking a development partner, we'd like to hear from you.
Get in Touch



